Molar weight
Calculate the weight of 2.00 moles of common salt (NaCl) in grams. M (NaCl) = 58.44 g/mol.
Final Answer:
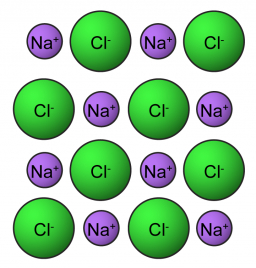
You need to know the following knowledge to solve this word math problem:
Units of physical quantitiesthemes, topicsGrade of the word problem
Related math problems and questions:
- Euclidean distance
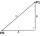 Calculate the Euclidean distance between shops A, B, and C, where: A 45 0.05 B 60 0.05 C 52 0.09 The first figure is the weight in grams of bread, and the second figure is the USD price.
Calculate the Euclidean distance between shops A, B, and C, where: A 45 0.05 B 60 0.05 C 52 0.09 The first figure is the weight in grams of bread, and the second figure is the USD price. - Crystal water
 The chemist wanted to check the water content of the crystallization of chromic potassium alum K2SO4 * Cr2 (SO4) 3 * 24 H2O, which took a long time in the laboratory. From 96.8 g of K2SO
The chemist wanted to check the water content of the crystallization of chromic potassium alum K2SO4 * Cr2 (SO4) 3 * 24 H2O, which took a long time in the laboratory. From 96.8 g of K2SO - Water material amount
 Calculate the material amount of a 1 kg water sample. M (H 2 O) = 18 g / mol.
Calculate the material amount of a 1 kg water sample. M (H 2 O) = 18 g / mol. - Spruce wood
 Calculate the weight of an edge made of spruce wood 6 m long when the cross-section of the edge is 146 cm square and if the density of the wood is 0.55 grams/cm cubic.
Calculate the weight of an edge made of spruce wood 6 m long when the cross-section of the edge is 146 cm square and if the density of the wood is 0.55 grams/cm cubic. - Salt solution
 Calculate the weight of a 50% salt solution we prepare from 120 g of salt.
Calculate the weight of a 50% salt solution we prepare from 120 g of salt. - Tin body mass
 A tin object transferred 150,000 J of heat, and its temperature dropped by 10 °C. Calculate the mass of the tin object. (c of tin is 0.227 kJ/kg. °C). Enter the resulting weight in grams and round it to two decimal places.
A tin object transferred 150,000 J of heat, and its temperature dropped by 10 °C. Calculate the mass of the tin object. (c of tin is 0.227 kJ/kg. °C). Enter the resulting weight in grams and round it to two decimal places. - Grams and kilos
 1. Tunji weighs 67 kg 114 g, while Deji weighs 54 kg 596 g. What is the sum total of their weight? 2. Adamu is 2 kg 324 g lighter than Muazu; if Muazu weighs 49 kg 158 g, what is Adamu's weight?
1. Tunji weighs 67 kg 114 g, while Deji weighs 54 kg 596 g. What is the sum total of their weight? 2. Adamu is 2 kg 324 g lighter than Muazu; if Muazu weighs 49 kg 158 g, what is Adamu's weight?
