Triangular prism
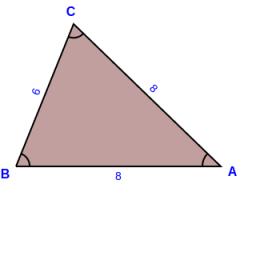
Calculate the surface of a triangular prism 10 cm high, the base of which is a triangle with sides of 6 cm, 8 cm, and 8 cm
Final Answer:
Tips for related online calculators
See also our trigonometric triangle calculator.
You need to know the following knowledge to solve this word math problem:
arithmeticsolid geometryplanimetryUnits of physical quantities
Related math problems and questions:
- Triangular prism
 Calculate the surface area and volume of a three-sided prism with a base of a right-angled triangle if its sides are a = 3 cm, b = 4 cm, c = 5 cm, and the height of the prism is v = 12 cm.
Calculate the surface area and volume of a three-sided prism with a base of a right-angled triangle if its sides are a = 3 cm, b = 4 cm, c = 5 cm, and the height of the prism is v = 12 cm. - Triangular RT prism
 Calculate the volume and surface of a triangular prism whose base is a right triangle with sides a = 3 m, b = ha = 4 m, and c = 5 m. The height of the prism is v = 5.5 m.
Calculate the volume and surface of a triangular prism whose base is a right triangle with sides a = 3 m, b = ha = 4 m, and c = 5 m. The height of the prism is v = 5.5 m. - Triangular prism
 Calculate the volume and surface of the triangular prism ABCDEF with the base of an isosceles triangle. Base's height is 16 cm, leg 10 cm, base height vc = 6 cm. The prism height is 9 cm.
Calculate the volume and surface of the triangular prism ABCDEF with the base of an isosceles triangle. Base's height is 16 cm, leg 10 cm, base height vc = 6 cm. The prism height is 9 cm. - Regular triangular prism
 Calculate the surface area of the body of a regular triangular prism when the length of its base edge is 6.5 cm, and its height is 0.2 m.
Calculate the surface area of the body of a regular triangular prism when the length of its base edge is 6.5 cm, and its height is 0.2 m. - Right triangle prism
 The lengths of the base legs are 7.2 cm and 4.7 cm, and the height of the prism is 24 cm. Calculate the volume and surface of a triangular perpendicular prism with the base of a right triangle.
The lengths of the base legs are 7.2 cm and 4.7 cm, and the height of the prism is 24 cm. Calculate the volume and surface of a triangular perpendicular prism with the base of a right triangle. - Triangular prism
 Calculate the surface of a regular triangular prism; the base's edges are 6 cm long, and the height of the prism is 15 cm.
Calculate the surface of a regular triangular prism; the base's edges are 6 cm long, and the height of the prism is 15 cm. - Triangular prism
 Calculate the surface of a triangular prism with the base of an equilateral triangle with a side length of 7.5 cm and a corresponding height of 6.5 cm. Prism height is 15 cm.
Calculate the surface of a triangular prism with the base of an equilateral triangle with a side length of 7.5 cm and a corresponding height of 6.5 cm. Prism height is 15 cm.
