Fuel efficiency
The gravimetric analysis of an anthracite fuel is 90% carbon, 3% hydrogen, 2% oxygen, and 1% nitrogen. Given that analysis of the dry combustion products by volume is 16.2% CO2, 3.5% O2, and 80.3% N2, during the combustion process of anthracite, which considers a solid fuel, some combustibles exit in fly ash with a weight fraction equal to 19% and temperature 270 °C, The fly ash is 15 % of the total ash, and there are losses due to combustibles in slag ash where the combustibles with weight fraction equal 5% and temperature 4500 °C. Ignoring the weight fraction of the water in the fuel, take the ambient condition 298 K, 0.1 MPa. Calculate the combustion efficiency if the weight fraction of ash in fuel is 30%. For anthracite fuel, take the value of the constant K in dry flue gas losses by 0.67 on the gross C.V basis and by 0.68 on the net C.V basis. The heat reaction of CO to that of CO2 equals 65.
Final Answer:

You need to know the following knowledge to solve this word math problem:
Units of physical quantitiesthemes, topicsGrade of the word problem
Related math problems and questions:
- Salami
 We have six kinds of salami, six of which have ten pieces, and one of which has four pieces. How many ways can we distinctly choose five pieces of salami?
We have six kinds of salami, six of which have ten pieces, and one of which has four pieces. How many ways can we distinctly choose five pieces of salami? - Petra
 Petra likes to bathe in a bathtub with a water temperature of 38 degrees. She turned on the hot water set at 70 degrees and left for a moment. Before she returned, 40 liters of water flowed into the tub. How much water with a temperature of 22 degrees mus
Petra likes to bathe in a bathtub with a water temperature of 38 degrees. She turned on the hot water set at 70 degrees and left for a moment. Before she returned, 40 liters of water flowed into the tub. How much water with a temperature of 22 degrees mus - Std-deviation
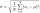 Calculate the standard deviation for file: 63,65,68,69,69,72,75,76,77,79,79,80,82,83,84,88,90
Calculate the standard deviation for file: 63,65,68,69,69,72,75,76,77,79,79,80,82,83,84,88,90 - Test scores
 Below is a collection of test scores from a class of 20 students. Make 2 histograms of the data. Choose your own horizontal scales as long as you have more than 4 cells in each histogram. 65 70 68 87 98 91 77 85 70 72 86 86 94 95 67 88 77 99 74 71
Below is a collection of test scores from a class of 20 students. Make 2 histograms of the data. Choose your own horizontal scales as long as you have more than 4 cells in each histogram. 65 70 68 87 98 91 77 85 70 72 86 86 94 95 67 88 77 99 74 71 - Heating Liquid Graph Analysis
 Colorless liquid weighing m = 200 g is heated with constant stirring on a stove with power input P0 = 600 W. 80% of the supplied energy is used to heat the liquid. Selected measured values of liquid temperature as a function of time are recorded in the ta
Colorless liquid weighing m = 200 g is heated with constant stirring on a stove with power input P0 = 600 W. 80% of the supplied energy is used to heat the liquid. Selected measured values of liquid temperature as a function of time are recorded in the ta - Electric cooker
 During which time does an electric cooker with power input P = 500 W and efficiency n = 75% heat water with mass m = 2 kg and temperature t1 = 10 °C to the boiling point (t2 = 100 °C)? The specific heat capacity of water is c = 4 180 J/kg/K.
During which time does an electric cooker with power input P = 500 W and efficiency n = 75% heat water with mass m = 2 kg and temperature t1 = 10 °C to the boiling point (t2 = 100 °C)? The specific heat capacity of water is c = 4 180 J/kg/K. - Heat energy
 Energy is measured using the unit Joule (J). Temperature is measured using the unit degree Celsius °C). To increase the temperature of 1 liter of water by 1 °C, 4200 J of heat energy is required. (a) Kelly wants to increase the temperature of 2 liters of
Energy is measured using the unit Joule (J). Temperature is measured using the unit degree Celsius °C). To increase the temperature of 1 liter of water by 1 °C, 4200 J of heat energy is required. (a) Kelly wants to increase the temperature of 2 liters of
